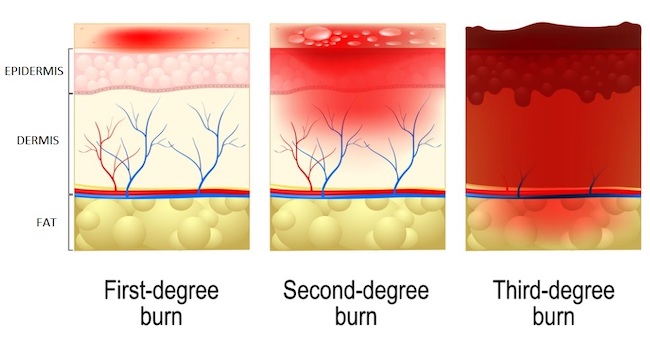
A severe burn injury is not static. Within 72 hours, partial thickness burns can progress, or convert, to full thickness burns, greatly increasing the risk of infection, incapacitating scarring, and even death.
Preventing the conversion is one of the most challenging aspects of treating burns, and a trans-institutional team of researchers from Vanderbilt University Medical Center and the School of Engineering has found a potential new approach—a prescription drug approved to treat inflammation in several autoimmune diseases, embedded and delivered in a topical gel, shows promise in reducing the severity of burn injuries.
“We have found that combining our microcapillary gel with immunosuppressants can prevent burn conversion in preclinical models,” said Wesley Thayer, associate professor of plastic surgery and vice chair of VUMC’s Department of Plastic Surgery. “This might prevent the need for surgery or even translate to lifesaving changes for large burns.

Current treatment typically involves use of topical antimicrobials for severe burns with full-thickness injury or, in less serious cases, the burn begins healing Most of the latter patients require surgery, and possibly skin grafts, to reconstruct the defect, Thayer said.
To develop a delivery method for the proposed treatment, he turned to Leon Bellan, associate professor of mechanical engineering, and his research group. Their work has included development of microfluidic networks in hydrogel materials and scalable micro/nanofabrication techniques for producing smart soft materials with novel functionality.

That team engineered a microcapillary hydrogel embedded with infliximab monoclonal antibodies. Infliximab, marketed under the brand Remicade, is used to treat rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, plaque psoriasis, and ulcerative colitis.
The results? The microcapillary gel, embedded with infliximab, reduced inflammation within partially burned skin and reduced progression to full-thickness burns. All subjects in the treatment cohort showed decreased conversion of burn from partial to full thickness injury when compared to the control group.
How about “Our microcapillary hydrogels are showing very promising results when delivering larger therapeutic molecules like infliximab to wound sites” Bellan said.
Severity of burns is determined by the percentage of burned area compared to the body surface area, the age of patient, and the depth of skin and soft tissue involvement. The presence of full-thickness burns, also known as third-degree burns, is associated with higher rates of physical deformities, limited movement and function, and incapacitating scarring as well as higher rates of mortality when compared to partial-thickness burns.
Because the inflammatory response may help drive burn conversion, the team wanted to determine whether infliximab, a powerful tumor necrosis factor inhibitor and anti-inflammatory drug, made a difference.
“Our study adds to the mounting evidence that TNF-a inhibition plays an important role in improving outcomes of wound healing, especially with burn injuries,” according to the researchers.
Infliximab can cost hundreds to even thousands of dollars for a single dose. Military personnel, industrial workers, and other populations at greater risk for severe burns may benefit the most from availability of such a treatment. The infliximab-laden gel also could be invaluable when burn injuries occur in combat or remote locations.
The research was published online in Burns.
Media contact
Pamela Coyle
School of Engineering